Nuclear quadrupole resonance is similar in concept, but unlike NMR it does not rely on the nuclei aligning themselves in an externally applied magnetic field. Rather than dealing with optical frequencies, NMR typically involves oscillations of just a few tens to hundreds of megahertz, which includes the band where broadcast FM radio stations operate.

In NMR, the energy difference between states is much less than for the electronic states of an atom, so the relevant frequencies are much lower. Conversely, if this excited-state electron falls back to the ground state, the atom will emit a photon of the exact same frequency to carry away the difference in energy. (It will be well defined as long as the applied magnetic field is uniform.) In that sense, the situation is much like that of an electron in an atom, which can be in the "ground" state or in a higher-energy "excited" state.Ī ground-state electron shifts to an excited state when the atom receives a dollop of electromagnetic radiation of just the right energy to put it there-that is, when it absorbs a photon of just the right frequency. The key is that the nuclei can occupy two distinct states separated by a well-defined increment in energy. But the magnetic moments of these nuclei, usually depicted as arrows, are allowed two possible orientations: in the same direction as the applied magnetic field or opposite to it.Īlthough alignment with the applied field is favored (this being the lower-energy condition), the energy difference between the two orientations is such that thermal agitation is usually sufficient to ensure that only slightly more than half the nuclei are in the lower-energy state. The laws of quantum mechanics dictate that when such nuclei are subjected to an externally applied magnetic field, they must align themselves along it. Nuclear magnetic resonance, first demonstrated in 1946, takes advantage of the fact that certain atomic nuclei possess magnetic dipole moments-that is, these nuclei act like tiny bar magnets, each with a north magnetic pole at one end and a south magnetic pole at the other. Nuclear quadrupole resonance has much in common with nuclear magnetic resonance (NMR), the fundamental physical process that makes magnetic resonance imaging possible. To understand why not, it is helpful to review the basic physics behind this promising technique. Detection through NQR does not face these difficulties. And one of the most high-tech tactics tried so far-sensing the nitrogen in explosives using thermal-neutron analysis-has proved to have inadequate sensitivity and specificity. For example, the ability of bomb-sniffing dogs and vapor detectors to sense explosives is influenced by environmental factors such as wind and ground moisture also, these approaches can fail with an explosive that is hermetically sealed, as is the case for some types of land mines and could readily be arranged in a terrorist bomb.
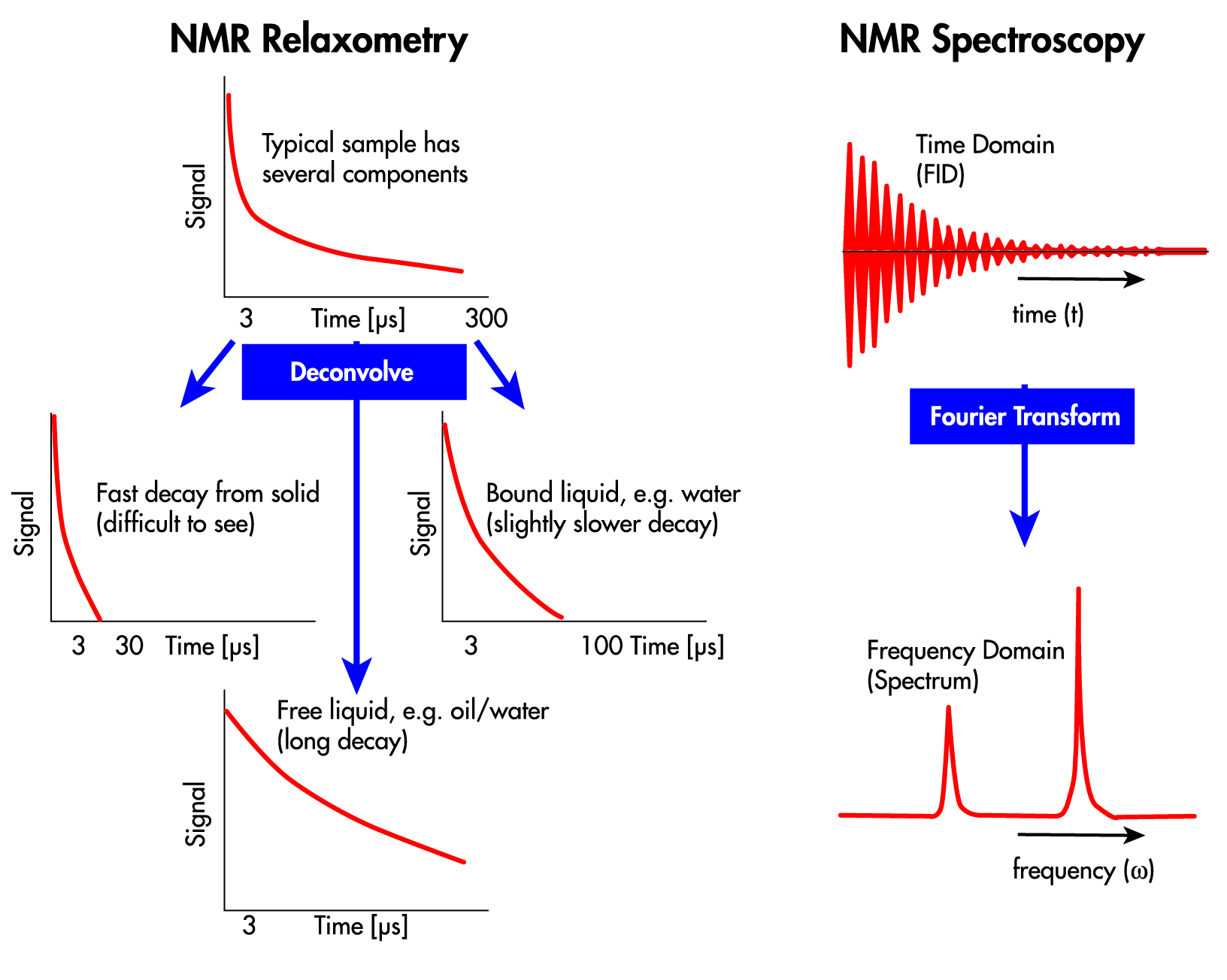
This approach offers some distinct advantages over the other options available. Specifically, we and our many government, academic and industry colleagues are trying to develop the means for detecting explosive chemicals based on a phenomenon called nuclear quadrupole resonance (NQR). We have been focused on that task for many years now.
